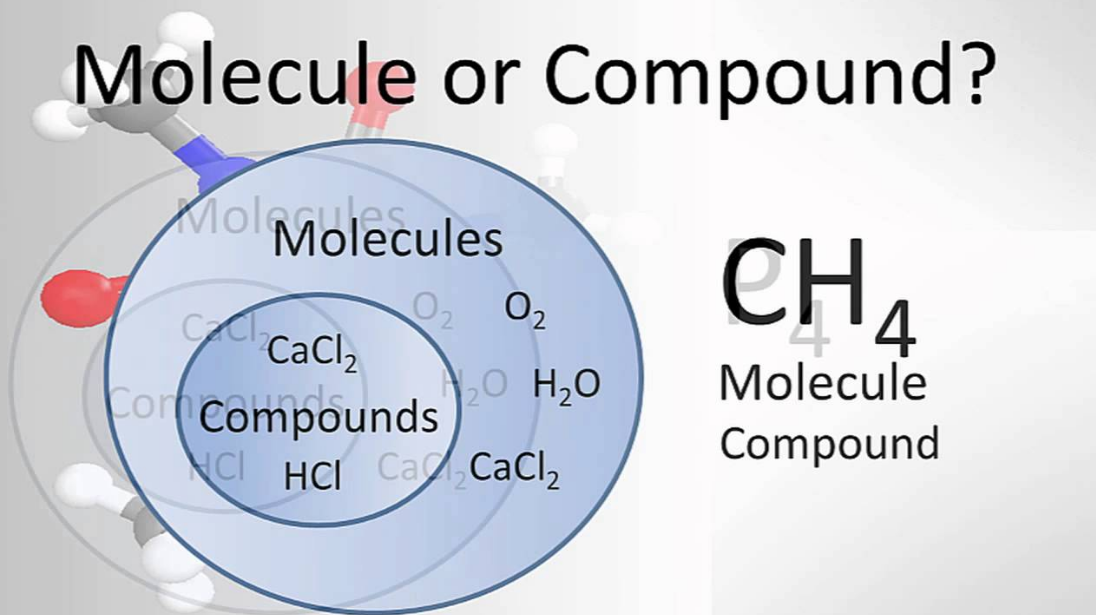
There is a great difference between molecule vs compound which must be known by everyone. Molecules are when the chemical bonds unite any atoms (two or more). On the other hand, compounds are molecules consisting of different atoms (two or more) of different chemical elements. So, you see that all compounds are molecules but not all molecules cannot be called compounds. In simpler terms, any combination of atoms is a molecule.
However, when the combination of atoms varies from elements, we refer to it as a compound. For instance, Hydrogen gas (H2), Oxygen (O2), water (H2O) are all molecules because they have two or more atoms. On the contrary, Calcium Carbonate (CaCO3), NaCl, H2O fall under the category of compounds because different elements with different atoms make them. Let us study in detail the two:
Table of Contents
Definition of Molecules
When two or more atoms come together in order to create the recognizable unit where the pure substance is able to retain its chemical properties plus composition despite dividing the unit, we refer to it as a molecule.
Moreover, molecules can either consist of atoms of the same element. On the other hand, they may also be of different chemical elements. If it comprises of the atoms from the same chemical element, we refer to it as a homonuclear such as Oxygen (O2).
However, if the molecule is made up of different atoms and elements, we refer to it as a heteronuclear. For instance, Water (H2O).
Definition of Compounds
Compounds are a group of different atoms and different elements which unite together by a chemical bond. The chemical bond can be of three types, either covalent, ionic, or metallic.
Similarly, it is important to remember that all compounds are molecules but not all molecules are compounds. Moreover, we only categorize heteronuclear molecules as compounds. However, the same is not the case with homonuclear molecules.
Therefore, we present compounds by their chemical formula. For instance, the chemical formula for water is H2O. Thus, it shows that the compound comprises 2 molecules of hydrogen and one molecule of oxygen.
More details about Molecules
There are different types of molecules that are based on different things. Further, the ones based on the number of atoms per molecule are Diatomic, Triatomic, and Polyatomic molecules.
Moreover, there are Homonuclear and Heteronuclear, Molecules. After that, there are organic and inorganic molecules. Finally, we have covalent and ionic molecules. Thus, they are based on the bond they share which can be either of the two.
Some examples are HCl, CO2, NO2, P4, C6H12O6, and more.
More details about Compounds
There are different types of compounds. Further, the ones which vary on the basis of the number of atoms present are diatomic, triatomic, and polyatomic. Moreover, the ones which we differentiate on the basis of complexity are simple and complex compounds.
Further, we have organic and inorganic compounds. Finally, there are covalent and ionic compounds. We differentiate them on the basis of covalent bonds or ionic bonds.
For instance, Acetic acid (C2H4O2), Ammonia (NH3), H2O, Hydrogen Peroxide (H2O2), NaCl, and more are examples of compounds.
FAQs
All these molecules are based on the number of atoms per molecule.
The difference between molecules and compounds is that molecules can be unstable whereas compounds are always stable.